The federal Drug Enforcement Administration (DEA) has announced it will soon be rolling out new forms for additional types of state-licensed medical marijuana businesses beyond dispensaries that want to take advantage of federal protections in line with the Trump administration’s cannabis rescheduling process.
Last month, DEA launched a registration form for medical marijuana dispensaries only, raising questions about how operators elsewhere along the cannabis supply chain could become federally compliant during a 60-day registration window for expedited processing that is scheduled to close on June 26.
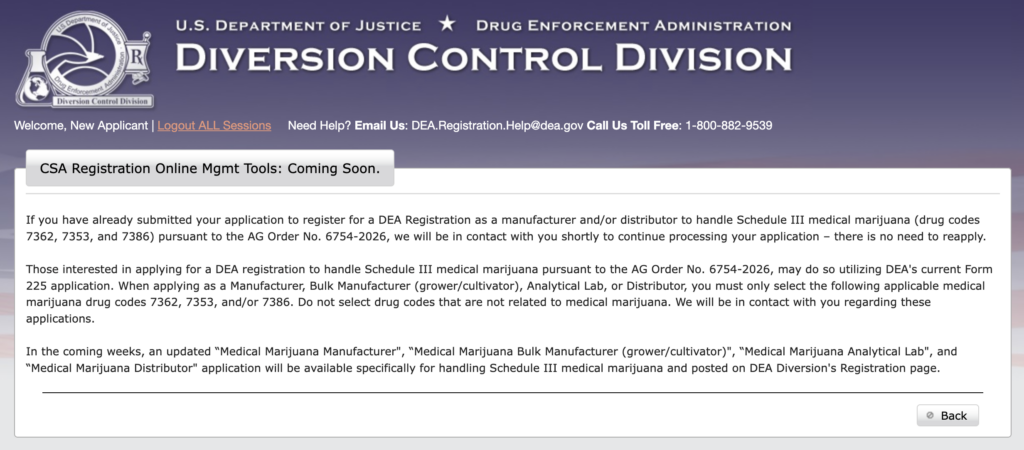
But now, the agency says that “in the coming weeks, an updated ‘Medical Marijuana Manufacturer’, ‘Medical Marijuana Bulk Manufacturer (grower/cultivator)’, ‘Medical Marijuana Analytical Lab’, and ‘Medical Marijuana Distributor’ application will be available specifically for handling Schedule III medical marijuana and posted on DEA Diversion’s Registration page.”
It’s not clear exactly how soon those forms will be posted, however.
In any case, the various DEA cannabis business registration portals are being launched following last month’s announcement by the Department of Justice that marijuana is being moved from Schedule I to Schedule III of the Controlled Substances Act (CSA), in phases.
Under an order signed by Acting Attorney General Blanche, marijuana products regulated by a
Read full article on Marijuana Moment


